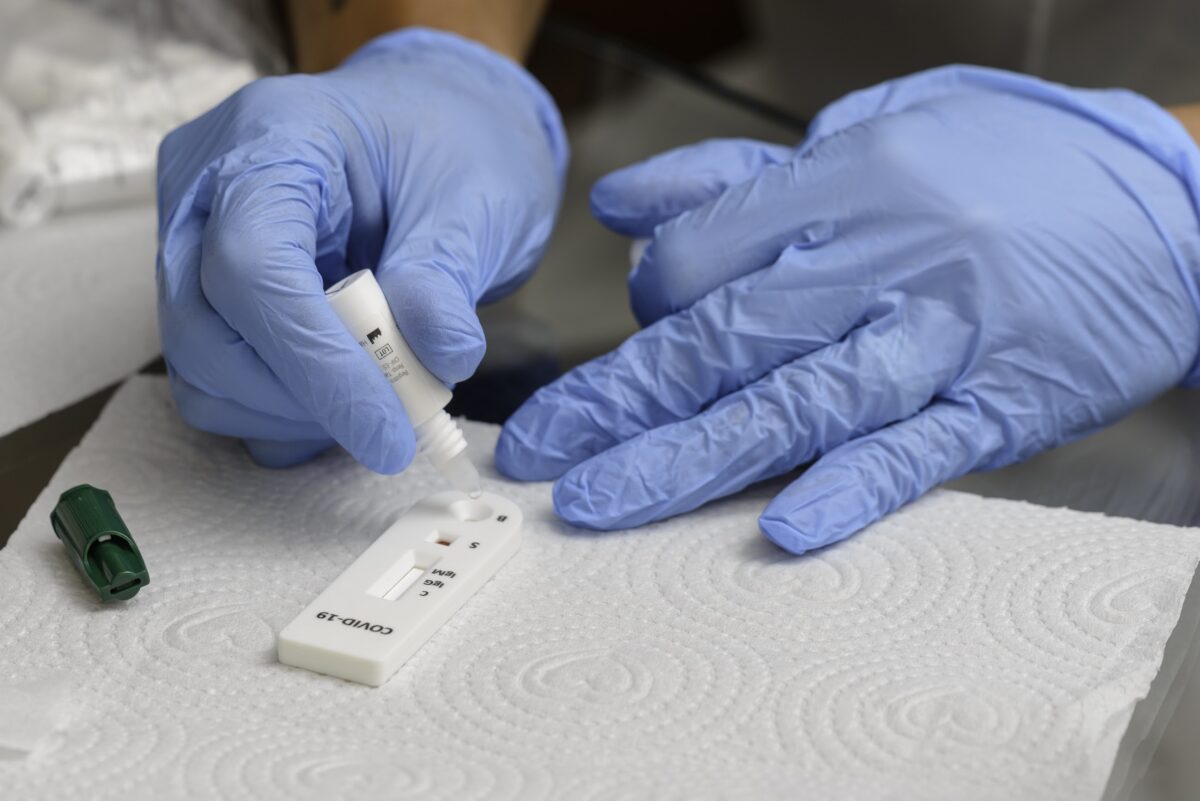
Participants identify key questions to take technical and programmatic research forward.
ACT-Accelerator diagnostic pillar partners joined in a virtual forum to discuss the development of a collaborative operational research agenda for SARS-CoV-2 antigen rapid diagnostic tests (Ag-RDTs).
The forum, held on 29 October 2020, was organized around three breakout groups according to three thematic areas for SARS-CoV-2 Ag-RDTs:
- Technical research
- Programmatic research
- Modelling
Group participants focused on understanding the performance of Ag-RDTs when implemented in real-world settings and across specific populations.
Priority questions
- Performance
- Use cases
- Samples
- Bio-safety
- Quality control
- Training and delivery
- Economics and cost.
Participants discussed steps to advance the Ag-RDT research agenda and agreed that there should be multiple future discussions on this.